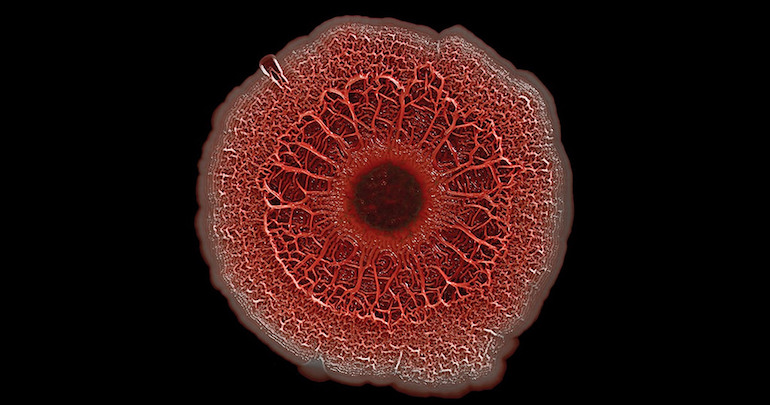
When a group of microorganisms needs to stick together, they build a biofilm. The cells cement themselves together onto a surface, forming durable structures that are notoriously hard to remove. In a medical setting, biofilms can contribute to dangerous antibiotic resistance. In the August issue of G3, Cromie et al. use a yeast model to identify six genes involved in biofilm formation, providing new leads for methods to combat biofilm-forming pathogens.
When forming a biofilm, microorganisms have to transition from a solitary lifestyle to one anchored down and surrounded by companions. This requires substantial changes in gene expression. To identify genes whose regulation contributes to biofilm formation, the authors used a genome-wide overexpression screen in a yeast strain that naturally forms biofilms. They examined 4,600 colonies that had been transformed with plasmids carrying individual yeast genes, looking for a change from the “fluffy” biofilm colony structure to the smooth unstructured globs typical of laboratory yeast. In the end, they identified six genes whose overexpression disrupted biofilm formation, causing colonies to change from fluffy to smooth.
Interestingly, all six of these genes are regulatory proteins—and four of them are transcription factors. To investigate the gene network changes triggered by overexpression, the authors performed RNA-seq on the six transformed yeast strains. They found evidence that the same transcriptional network was activated by overexpression in five of the six strains, and this network contains some genes already known to affect colony morphology.
Expression levels of these regulators may act as a switch that yeast that can use to easily transition between growing in a solitary state and growing as a biofilm. Targeting these switches might be one way to prevent pernicious biofilms from forming in the first place.
CITATION
Transcriptional Profiling of Biofilm Regulators Identified by an Overexpression Screen in Saccharomyces cerevisiae
Gareth A. Cromie, Zhihao Tan, Michelle Hays, Amy Sirr, Eric W. Jeffery and Aimée M. Dudley
G3: GENES, GENOMES, GENETICS August 1, 2017 vol. 7 no. 8 2845-2854; https://doi.org/10.1534/g3.117.042440